aim to act as an international group, supporting, informing and networking with anyone affected by a rare chromosome disorder or an autosomal dominant single gene disorder and with any interested professionals.
Iran rare diseases foundation get new membership from CMTC-OVM ORGANIZATION
CMTC-OVM is a worldwide non-profit community that aims to improve the quality of life of people suffering from vascular abnormalities (blood vessel abnormalities), such as CMTC, their families, and stimulate scientific research into these disorders.
Membership of Iran’s Rare Diseases Foundation in Austria’s Airways and Allergies Platform

It is hoped that with the cooperation of this organization, we will take steps towards the awareness and treatment of such rare diseases and patients.
What is a Rare Disease?
- By Dr. Priyom Bose, Ph.D.Reviewed by Emily Henderson, B.Sc.
Any unusual life-altering complication of the human body affecting a small number of people is classified as a rare disease (RD). These diseases are complex and possess a significant challenge both in terms of diagnosis and management. The field of rare diseases is constantly evolving.
A plethora of rare conditions are constantly being added to the already existing pool of rare complications. Due to this, clinicians and researchers across the globe are working tirelessly to create better management and therapeutic approaches for the patients.
Image Credit: valiantsin suprunovich/Shutterstock.com
Rare Disease (Looking Deeper)
Compared to the innumerable number of patients affected by prevalent diseases like diabetes, heart diseases, etc., a rare disease has a limited number of patients. So far, about 5000-8000 unique, rare diseases have been described. Rare diseases affect approximately 400 to 700 million individuals globally, making them a global healthcare issue.
An RD affects around 60 million individuals in the United States and the European Union. The number of RDs is currently estimated to be in the thousands. These conditions are frequently significant, harm one’s quality of life, and can even be fatal. Most RDs have a genetic component, with 72–80 % of these disorders having a gene or genes identified.
They are usually defined by their low frequency, measured in terms of prevalence or incidence within a country or geographical region. In the medical literature, five novels RDs are characterized on average every week. According to a global study of RD terminology, 58% of definitions contained a prevalence criterion, with a global average of 40 cases per 100,000 persons.
According to the European Medicines Agency or EMA, rare diseases can be described as a medical condition with a prevalence of fewer than 5 patients in 10,000 people. The research and clinical development surrounding rare diseases are escalating at a positive rate. However, there is a lot more to explore and understand.
The Various Challenges
RDs encompass a wide range of disorders and clinical manifestations, however, the vast majority of RDs afflict children and are chronic and life-threatening. The majority of rare diseases aren’t curable, with only a few having efficient available therapies. The recognition and diagnosis of RDs present several interrelated issues. They significantly impact the quality of epidemiologic and clinical investigations and complicate the definition of unmet patient requirements, safety, possible efficacy, effectiveness, and value of RD treatments.
Inadequate awareness and knowledge of RDs can lead to misdiagnosis and a delay in receiving a definitive diagnosis. Patients who are unfamiliar with relevant signs and symptoms may fail to seek medical help when necessary. Similarly, professionals may misdiagnose the disease or mistake symptoms for those of more frequent diseases with which they are more familiar. The comparability of data from epidemiologic and clinical investigations may be significantly limited if we don’t fully grasp the disease and its cause.
Heterogeneity in pathogenesis, presentation of signs and symptoms, disease severity, and development might make it difficult to characterize a rare disease in clinical research and detect it in ordinary clinical practice, leading to misdiagnosis and underestimating of genuine disease frequency.
In patients with pathognomic clinical characteristics, diagnosing an RD may be simple. Diagnostic certainty, on the other hand, might be a challenge due to the variety of many RDs and the difficulty in correctly interpreting sophisticated investigative testing methods.
Biotechnology eBook Compilation of the top interviews, articles, and news in the last year.
Misdiagnoses are widespread as a result of the aforementioned issues, which can lead to ineffective therapy. Such medication may not only cause adverse effects, but it may also hide signs of the underlying problem, delaying the accurate diagnosis and beginning of suitable treatment even longer. An RD’s occurrence varies by geography and country, ranging from dense clustering in certain areas to vast distribution in others. In the situation of prevalence heterogeneity, prevalence statistics from epidemiologic studies undertaken in a specific region may be incorrectly extrapolated to larger geography.
Overcoming the hurdles
Rare disease research has received a great deal of attention in recent decades. It is primarily due to legislation designed to make it easier for patients to have access to effective treatments by rewarding pharmaceutical and biotechnology companies to develop novel medications that would otherwise be unprofitable. In the field of rare diseases, there has also been a remarkable increase in social awareness and worldwide connectedness. Several groups are forming to provide information to the general public and to educate them about uncommon diseases.
As a result, governments all over the world have realized the importance of enacting laws and regulations to encourage the development of new and novel technology for the treatment of RDs. The detection of RDs has improved due to advancements in molecular genetics, disease pathophysiology, and medical technology. They’ve also aided researchers in figuring out how to improve the diagnosis, prognosis, treatment for rare diseases and precisely subclassify common diseases into groups of RDs with different characteristics.
Patient Registries for Rare Diseases: A Goldmine
Patient registries are a valuable instrument in the field of rare diseases (RD) that aids clinical research, promotes the development of appropriate clinical trials, improves patient care, and supports healthcare administration. They create a critical information infrastructure that supports the work of European Reference Networks (ERNs) on rare diseases.
There has been a significant expansion of RD registries in recent years, necessitating the development of guidance for the basic requirements, recommendations, and standards required to maintain a high-quality registry. Several such registries have been formed in recent years including, the Registry for Neuromuscular Disease, Central Registry of Rare Diseases (CRRD), Ataxia-Telangiectasia International Registry, EHDN: registry of juvenile Huntington’s disease, and EUROCARE CF: European cystic fibrosis registry.
The Rare Diseases Registry Program (RaDaR) website was developed in 2021 by NCATS to give the rare disease community free and easily available instructions on how to set up and manage high-quality registries. RaDaR will be a dynamic website. In a staged approach, NCATS staff will continue to add new instructions, best practices, testimonials, and other resources from the rare disease community to the site. Although the researchers exploring the area of rare diseases have achieved significantly, we still have a long road ahead of us.
References:
- Navarrete-Opazo, A. A., Singh, M., Tisdale, A., Cutillo, C. M., & Garrison, S. R. (2021). Can you hear us now? The impact of health-care utilization by rare disease patients in the United States. Genetics in medicine: official journal of the American College of Medical Genetics, 23(11), 2194–2201. https://doi.org/10.1038/s41436-021-01241-7
- Nestler-Parr, S., Korchagina, D., Toumi, M., Pashos, C. L., Blanchette, C., Molsen, E., Morel, T., Simoens, S., Kaló, Z., Gatermann, R., & Redekop, W. (2018). Challenges in Research and Health Technology Assessment of Rare Disease Technologies: Report of the ISPOR Rare Disease Special Interest Group. Value in health: the journal of the International Society for Pharmacoeconomics and Outcomes Research, 21(5), 493–500. https://doi.org/10.1016/j.jval.2018.03.004
- Kodra, Y., Weinbach, J., Posada-de-la-Paz, M., Coi, A., Lemonnier, S. L., van Enckevort, D., Roos, M., Jacobsen, A., Cornet, R., Ahmed, S. F., Bros-Facer, V., Popa, V., Van Meel, M., Renault, D., von Gizycki, R., Santoro, M., Landais, P., Torreri, P., Carta, C., Mascalzoni, D., … Taruscio, D. (2018). Recommendations for Improving the Quality of Rare Disease Registries. International journal of environmental research and public health, 15(8), 1644. https://doi.org/10.3390/ijerph15081644
- Richter, T., Nestler-Parr, S., Babela, R., Khan, Z. M., Tesoro, T., Molsen, E., Hughes, D. A., & International Society for Pharmacoeconomics and Outcomes Research Rare Disease Special Interest Group (2015). Rare Disease Terminology and Definitions-A Systematic Global Review: Report of the ISPOR Rare Disease Special Interest Group. Value in healt : the journal of the International Society for Pharmacoeconomics and Outcomes Research, 18(6), 906–914. https://doi.org/10.1016/j.jval.2015.05.008
- Griggs, R. C., Batshaw, M., Dunkle, M., Gopal-Srivastava, R., Kaye, E., Krischer, J., Nguyen, T., Paulus, K., Merkel, P. A., & Rare Diseases Clinical Research Network (2009). Clinical research for rare disease: opportunities, challenges, and solutions. Molecular genetics and metabolism, 96(1), 20–26. https://doi.org/10.1016/j.ymgme.2008.10.003
- RaDaR. [Online] National Center for Advancing Translational Sciences. Available at: https://ncats.nih.gov/radar
Last Updated: Feb 15, 2022
UB research identifies key mechanism in infantile cystinosis
University at Buffalo research has identified how a misstep in the genesis of a key component of the kidney causes infantile cystinosis, a rare disease that significantly shortens the lifespan of patients. Published Nov. 30 in the International Journal of Molecular Sciences, the work reveals that the mechanisms that cause the disease could be addressed and potentially cured through the genome-editing technique CRISPR. That could make kidney transplants, the most effective treatment currently available for these patients, unnecessary.
Infantile cystinosis, the most common and most severe type of cystinosis, occurs as the result of an accumulation in the body’s cells of cystine, an amino acid. The buildup damages cells throughout the body, especially the kidneys and the eyes. Treatment consists of medications that work to lower the level of cystine in the body, as well as therapies that address the impaired growth of these children due to the inability to properly absorb nutrients. Some children require feeding tubes. Eventually, patients with infantile cystinosis, also called nephropathic cystinosis, will require dialysis and a kidney transplant.
Promise of stem cells
Human-induced pluripotent stem cells (hiPSCs) are stem cells that can differentiate into many different cell types. They hold tremendous potential for studying genetic diseases; the drawback has been that differentiation into certain cell types has been problematic. Such is the case with many cell types found in the kidney.
But a new protocol developed by this research team was successful.
Ramkumar Thiyagarajan, PhD, assistant professor of geriatric studies at the University of Kansas and formerly a postdoctoral fellow at UB, is first author on the paper.
The protocol involved extracting stem cells from a healthy individual and an individual with infantile cystinosis. The researchers developed a culture medium to grow stem cells that included a small number of defined components present in blood, including insulin, specific proteins, growth factors and others. “Conducting the differentiation protocol under these conditions occurred in a timely manner,” says Taub, “we didn’t have to wait for weeks on end, and it occurred in a reproducible manner.”
The researchers were able to efficiently differentiate the hiPSCs into the kidney proximal tubule, the type of nephron in the kidney that is impaired in infantile cystinosis, as well as in other kidney diseases.
“Unlike in other studies, we were able to retain a number of markers in the tubule that are physiologically important in the kidney’s reabsorptive functions,” says Taub. “Although these markers were expressed in both the normal and the cystinosis-derived hiPSCs, the genesis of the tubule was impaired in the cystinosis-derived cells, mimicking what happens in infantile cystinosis.”
A potential cure
That finding means that the CRISPR genome-editing technique could be used to repair the defective genome and potentially cure the disease. “The normal gene can be introduced in the genome of cystinotic hiPSCs, which can then be injected in the kidney to replace the defective proximal tubules of individuals with infantile cystinosis,” Taub says.
“In cystinotic individuals, it is the renal proximal tubule that degenerates, presumably due to programmed cell death,” explains Taub, “so the entire kidney would not need to be replaced. The defective renal proximal tubules of individuals with this disease can be replaced with normal tubules following the introduction of the normal gene into cystinotic hiPSCs obtained from the patient. And because these tubules are from cells derived from the patient, there should be no problem with tissue rejection.”
The findings are applicable to other kidney diseases where the renal proximal tubule is damaged, including acute kidney injury that can lead to chronic kidney disease and renal failure, and can be fatal.
Initial studies will need to be conducted with animal models as well as with in vitro tissue culture cells.
The research was funded by UB’s WNYSTEM and The Cystinosis Research Foundation.
Source:
Journal reference:
Thiyagarajan, R & Taub, M. (2023). Studies with Human-Induced Pluripotent Stem Cells Reveal That CTNS Mutations Can Alter Renal Proximal Tubule Differentiation. International Journal of Molecular Sciences.
National Strategic Plan for Rare Diseases of Iran


The providing and editing of the national rare diseases document is one of the most vital activities of the Rare Diseases Foundation, and in this regard, the Rare Diseases Foundation of Iran, with a lot of endeavor and follow-up, and with the participation and cooperation of the professors and expertise of the Foundation and other universities of medical sciences, prepared and compiled the National Document of Rare Diseases of Iran, which after being approved by the Secretariat of the Supreme Council of Health and Food Safety and the cooperation and collaboration of the honorable officials of that ministry and other related institutions, was finally approved by the honorable president and announced. Undoubtedly, the declaration of this document can be a very huge and effective step in determining the duties of the institutions and beneficiaries related to these patients, providing services to rare patients and their families, and solving the problems of these patient respectfully


















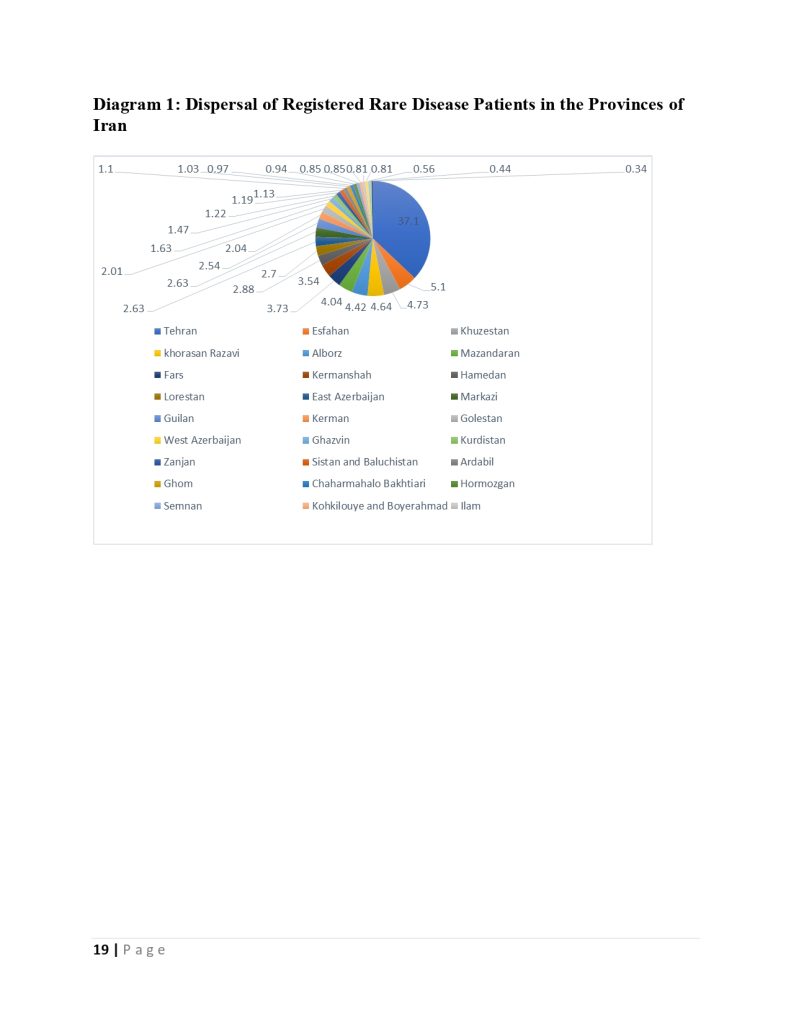
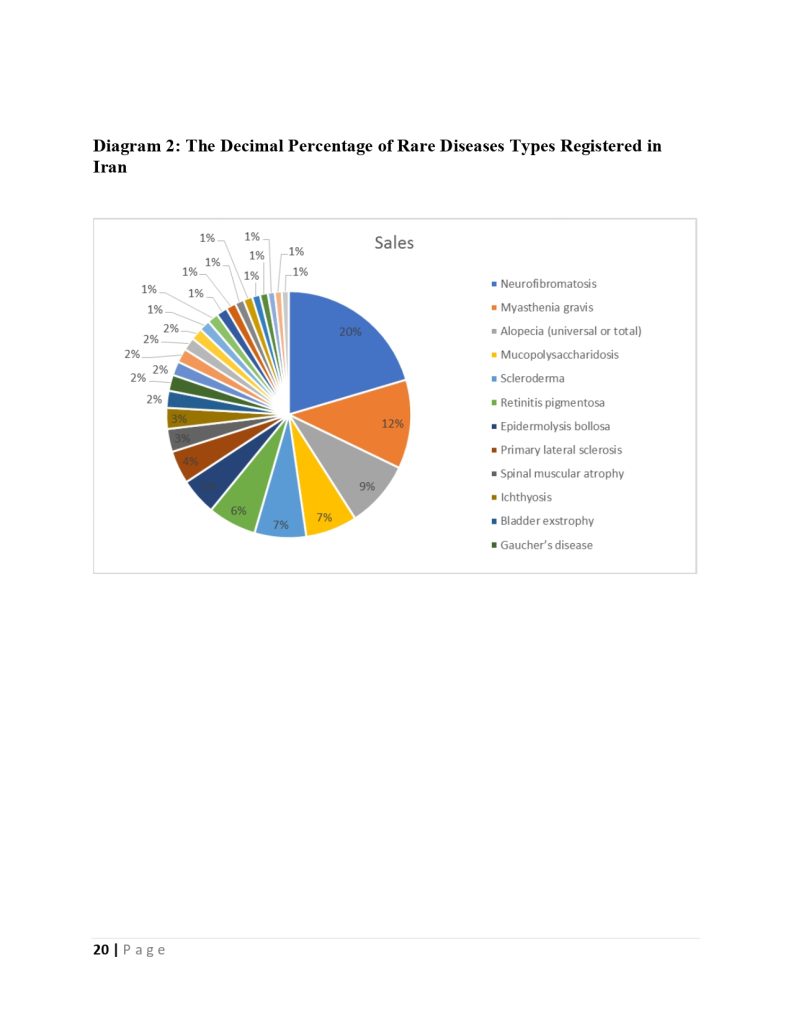
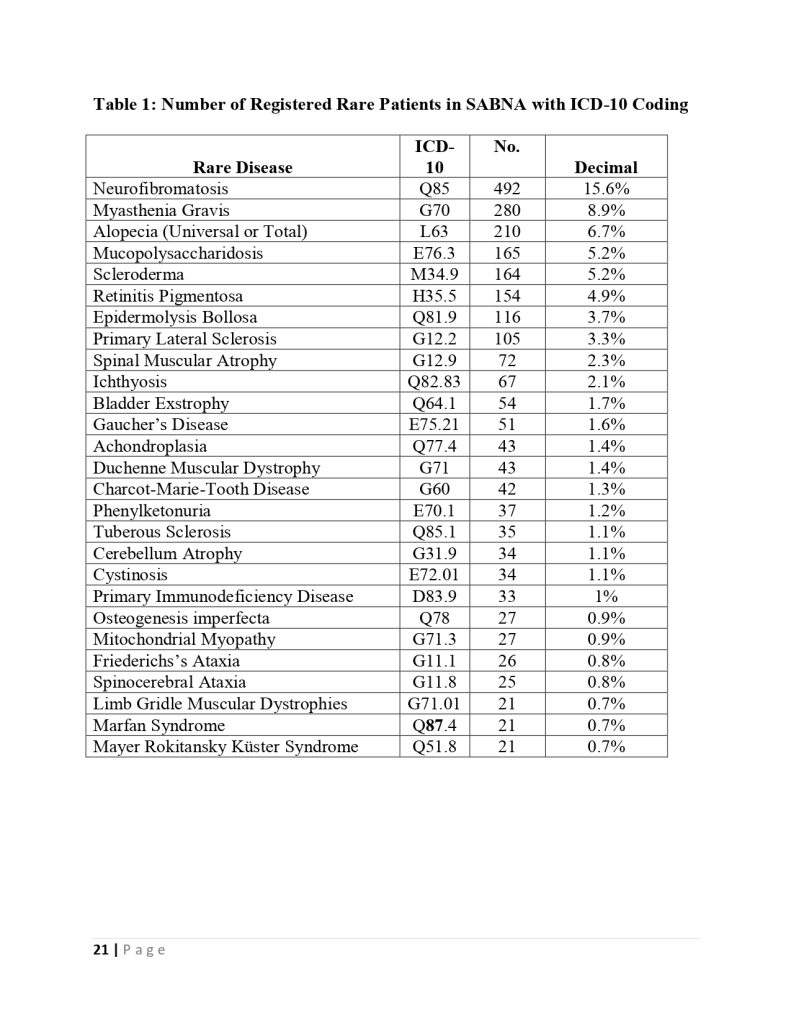






















There are 63 service packages in the fund for rare and hard-to-treat patients

The deputy of insurance and services of the iran health insurance organization declared :we cover one million and thousand patients in the fund for rare and hard treatment diseases.
In report of mehr news agency ,Mehdi rezaei,
According the insurance coverage of rare and hard treatment patients, announce : 1,600 billion tomans have been paid in the first 4 months of this year to cover the costs of rare and hard treatment patients .
Most of services are provided in the pharmaceutical, medical radiation and laboratory services, 75% of which are related to the pharmaceutical sector .
Rezaei added: 63 service packages have been prepared in this fund for these patients, who have received the most services in the rare and hard treatment patients fund, respectively , for cancer , MS, AND HEMOPHILIA