RADOIR’s first medical committee was held in 2020 with delay due to COVID-2019 pandemic.
FDA Drug Approvals Report for 2019 New Treatments for Rare Diseases
The FDA’s Center for Drug Evaluation and Research (CDER) released the annual report, “Advancing Health Through Innovation:
New Drug Therapy Approvals”, outlining new drugs approved for the first time.
FDA Drug Approvals Report for 2019 New Treatments for Rare Diseases
The FDA’s Center for Drug Evaluation and Research (CDER) released the annual report, “Advancing Health Through Innovation:
New Drug Therapy Approvals”, outlining new drugs approved for the first time.
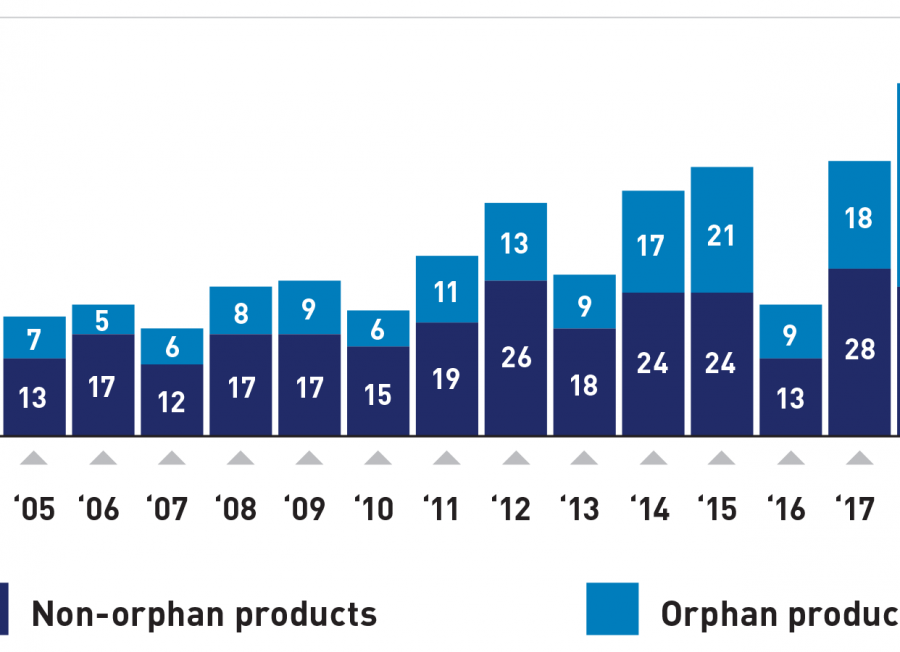
Included in the report were numerous approvals for treatments targeted at combatting rare diseases like cystic fibrosis, tenosynovial giant cell tumor, and sickle cell disease. Additional approvals were given to assist patients with erythropoietic protoporphyria, neuromyelitis optica spectrum disorder, systemic sclerosis-associated interstitial lung disease, and Duchenne muscular dystrophy. Because so few patients suffer from these diseases, there are often scarce or no drugs available to treat the conditions.
2019Golden Rose as Glorious Medicine Award For RADOIR’s Managing Director
Dr. Hamid Reza Edraki, scientific board member of SBMU & RADOIR’s managing director was awarded the Golden Rose for his glorious scientific activities and impressive medical services.
RDI signs Memorandum of Understanding with the World Health Organization
RDI has signed a collaboration agreement with WHO to start the journey towards a global network of centers of excellence for rare diseases
The Head of the MOH-Iran’s Management Center for Transplantation and Special Diseases Conferred on The Status of Rare Patients in Spain
Dr. Mahdi Shadnoush, the head of management center for transplantation and special diseases at the Iranian Ministry of Health had a meeting session in Dec.2019 about the status of rare patients at the Spanish Rare Diseases Research Institute in Spain as per the follow ups and arrangements done by Rare Diseases Foundation of Iran.
“Fedratinib” Approved by FDA For Forms of “Myelofibrosis”
FDA approved “Fedratinib” (Inrebic) capsules for rare adult patients with special forms of Myelofibrosis.
Triple Therapy to Treat Cystic Fibrosis Approved by FDA
Trikafta (elexacaftor/ ivacaftir/tezacagtor) as the first triple combined therapy for CF mutation has been approved.
EURORDIS strongly urges EU Council to preserve a HTA legislation that best serves patients
Rare Diseases Europe, an alliance of over 800 rare disease patient organisations, is today urging Member States of the EU to take off the table the proposal of the Presidency of the EU Council to withdraw Article 7 of the current proposal for European cooperation on health technology assessment (HTA).